 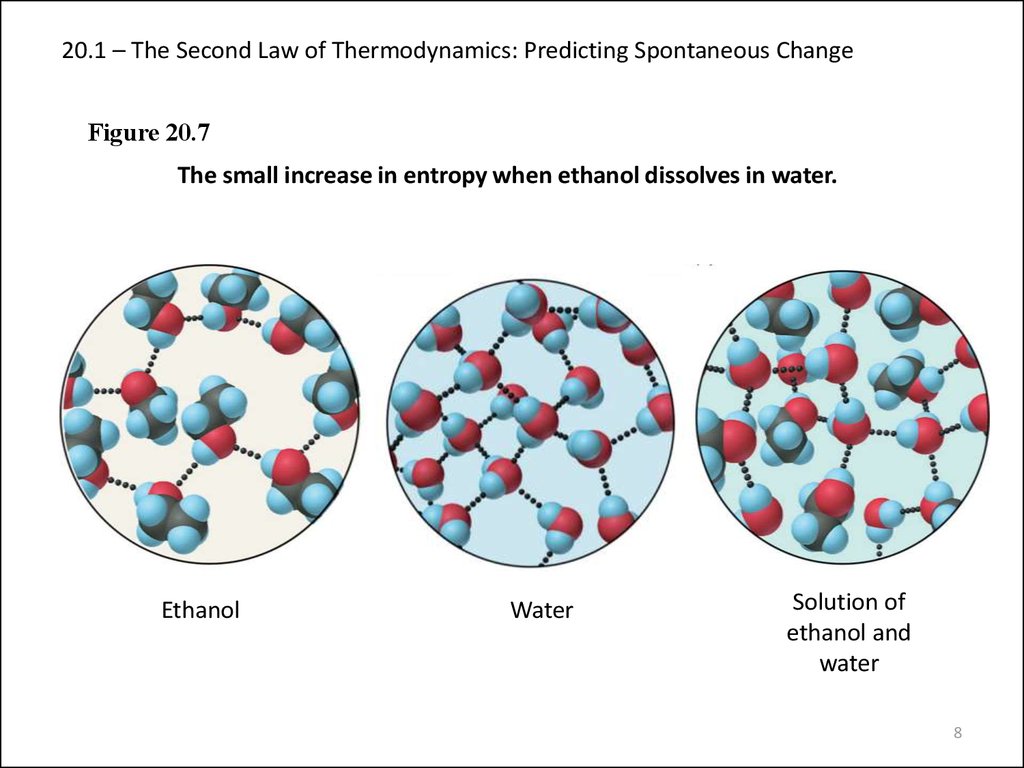
The third law of thermodynamics can be stated as:Ī system's entropy approaches a constant value as its temperature approaches absolute zero. Natural processes tend to go only one way, toward less usable energy and more disorder. The second law of thermodynamics states that the entropy in an isolated system always increases. It states that, in an isolated system, entropy can increase but cannot decrease. What are the 1st and 2nd laws of thermodynamics The first law of thermodynamics also known as the law of conservation of energy states that energy can neither be created nor destroyed, but it can be changed from one form to another. A) Living organisms do not obey the second law of thermodynamics, which states that entropy must increase with time. The second law of thermodynamics says that some things can't be undone after they are done. The energy gained (or lost) by a system is equal to the energy lost (or gained) by its surroundings. 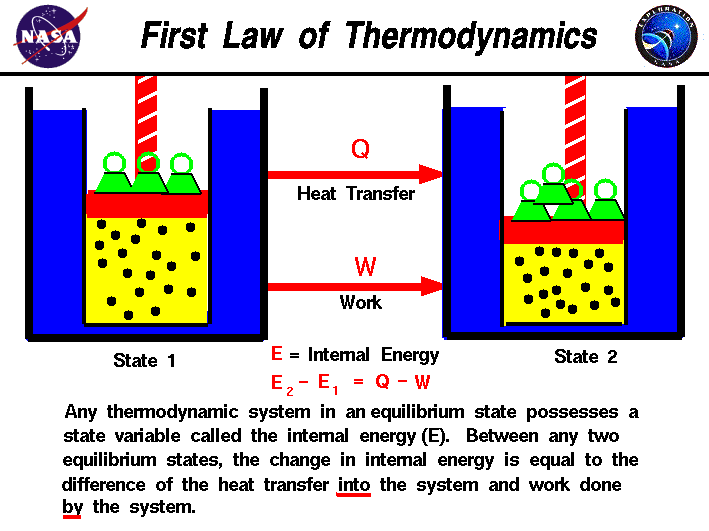
They are used in thermodynamics and other sciences, for example chemistry. It can also be stated in the following form: They talk about temperature, heat, work, and entropy. The law of conservation of energy states that the total energy of an isolated system is constant energy can be transformed from one form to another, but can be neither created nor destroyed. The first law of thermodynamics is a version of the law of conservation of energy, adapted for thermodynamic systems. 1 Application of the second law of thermodynamics helps explain the various ways in which engines transform. If two systems are both in thermal equilibrium with a third system, then they are in thermal equilibrium with each other. The second law of thermodynamics states that you can move heat from a hotter place to a colder place without doing work, but that you need to work to move heat from a colder place to a hotter place. It states the transitive relationship between the temperatures of multiple bodies in thermal equilibrium. The zeroth law of thermodynamics says temperature is an empirical parameter in thermodynamic systems. Then there was another law, called the "zeroth law." The law of conservation of mass is also an important idea in thermodynamics, but it is not called law. Thermodynamics has three main laws: the first law, the second law, and the third law. Angle Sum Formula Study with Quizlet and memorize flashcards containing terms like Sin (2 delta ), Cos (2 delta). They are used in thermodynamics and other sciences, for example chemistry. The second law of thermodynamics states that the entropy (spontaneity) of an isolated system will increase over time. 
The second law also states that the changes in the entropy in the universe can never be negative. They talk about temperature, heat, work, and entropy. The Second Law of Thermodynamics states that the state of entropy of the entire universe, as an isolated system, will always increase over time. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
